Oxygenation of RZn(N,O)-type complexes as an efficient route to zinc alkoxides not accessible via the classical alcoholysis path
Zbigniew Wróbel,a Tomasz Pietrzak,b Iwona Justyniak,a Janusz Lewiński,ab
aInstitute of Physical Chemistry, Polish Academy of Sciences, Kasprzaka 44/52, 01-224 Warsaw, Poland
bFaculty of Chemistry, Warsaw University of Technology, Noakowskiego 3, 00-664 Warsaw, Poland
DOI: 10.1039/C7CC05818G
First published online 12 September 2017.
Paper at Publisher's website
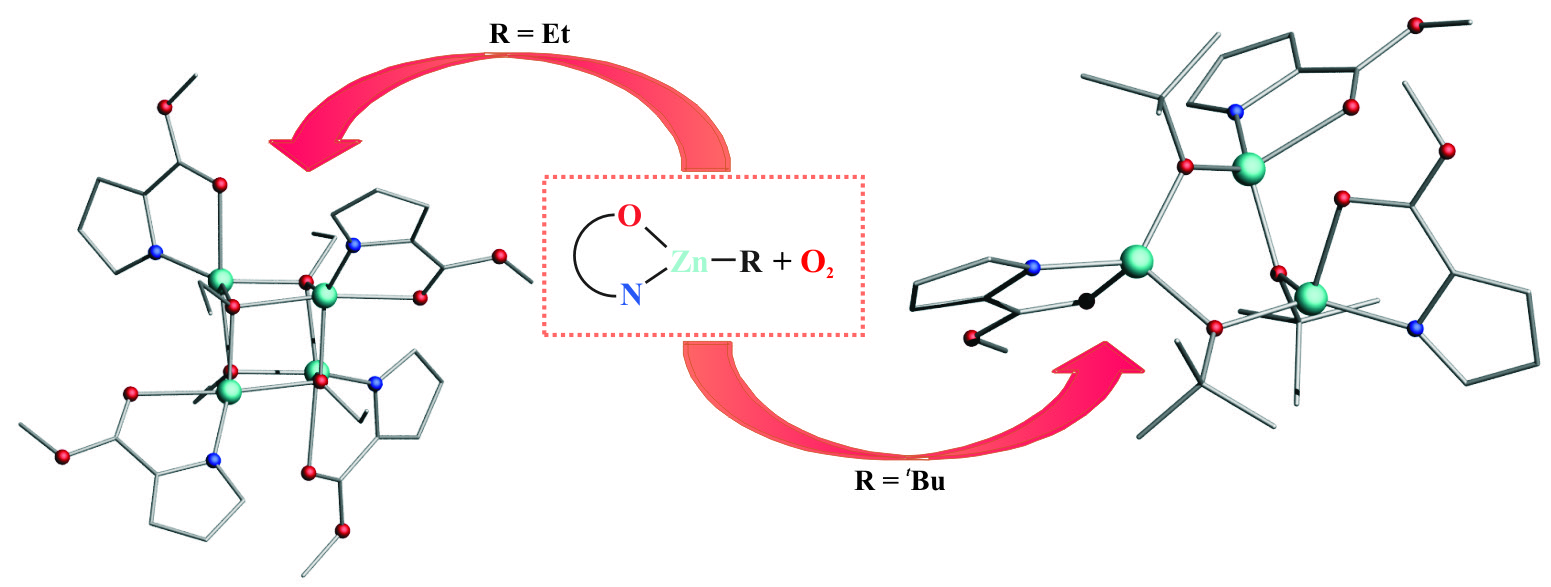
The controlled oxygenation of alkylzinc complexes supported by a 2-ester substituted pyrrolate ligand (L) leads to zinc alkoxides with an uncommon structural motif in the solid state: a trimer [(L)Zn(μ-OtBu)]3 with the central [Zn3(μ-OR)3] ring and a tetramer [(L)Zn(μ3-OEt)]4 with a heterocubane-type structure. Strikingly, these seemingly simple zinc alkoxides are not accessible via the classical alcoholysis route.